 FOCS-1, a continuous cold caesium fountain atomic clock in Switzerland, started operating in 2004 at an uncertainty of one second in 30 million years. The final data column compares this percentage against the percentage of all minerals that contain the element listed in each row. Covalent radius: 244☑1 pm: Van der Waals radius: 343 pm: Spectral lines of caesium: Other properties. 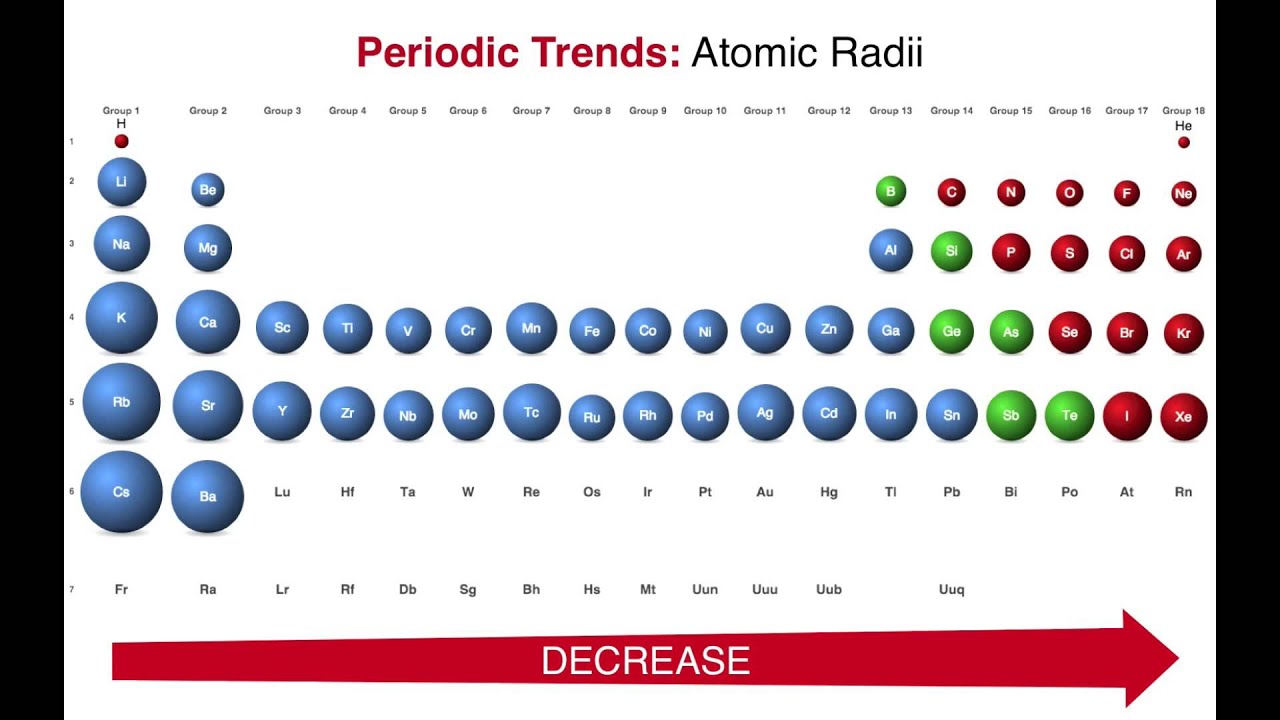
The second data column lists this number as a percentage of all minerals listed with Caesium. But caesium is in the 6th period of the periodic table whereas chlorine is in the 3rd. How many cesium atoms would have to be laid side by side to give a row of cesium atoms 1.00 in. The first data column contains the total number of minerals listed with Caesium and the element listed for that row. My book mentions that the cation to anion radius ratio in caesium chloride is about 0.93, and since this number lies between 0.732 and 1, it suggests a cubic arrangement with a coordination number of 8. The radius of a cesium atom is 2.65 Angstroms. It must be noted, atoms lack a well-defined outer boundary. Note that unlike other sections on this page this includes non-essential elements. The atomic radius of Caesium atom is 244pm (covalent radius). The atomic radius of Caesium atom is 244pm (covalent radius). (b) The metallic atomic radius, rmet, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum. (a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. Caesium is highly ionic in character and the ionic radius of Cs 1+ is 188 pm in 12-fold coordination.  
The lone 6 s electron is relatively easily removed this first ionization potential of Cs is 375.4 kJ/mol. This table compares the known valid mineral species listed listed with Caesium and the other elements listed based on the official IMA formula. Figure 8.6.2: Definitions of the Atomic Radius. Caesium, atomic number 55, has a standard atomic weight of 132.90545 and an electronic configuration of Xe6 s 1. 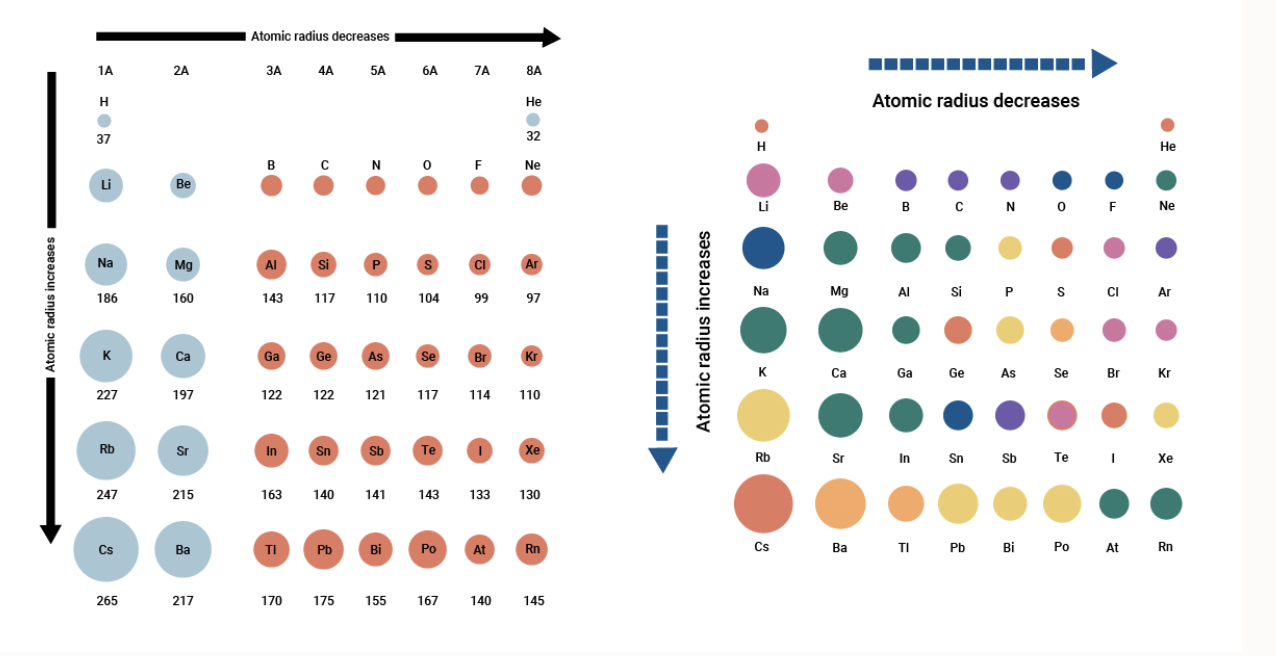
Its crystal structure forms a major structural type where each caesium ion is coordinated by 8 chloride ions. This colorless salt is an important source of caesium ions in a variety of niche applications. Minerals where Cs was previously thought to be a colouring agent probably contain other ions such as Mn#2+#.Ģ7 valid species containing essential CaesiumĬs + enters later phases in igneous rocks due to its large size.Ītom mole fraction relative to Si=1 (% uncertainty)Įlement association of Caesium in the Mineral World Caesium chloride or cesium chloride is the inorganic compound with the formula Cs Cl. In keeping with overall periodic trends, the atomic and ionic radii increase smoothly from Li to Cs, and the first ionization energies decrease as the atoms become larger.Wikipedia WebElements Los Alamos National Laboratory Theodore Gray's Ĭaesium as a chromophore in minerals and gemsĭoes NOT cause colour as it has no electrons in d-orbitals. The variant spelling cesium is sometimes used, especially in North American English, but caesium is the spelling used by the IUPAC, although since 1993 it has. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
